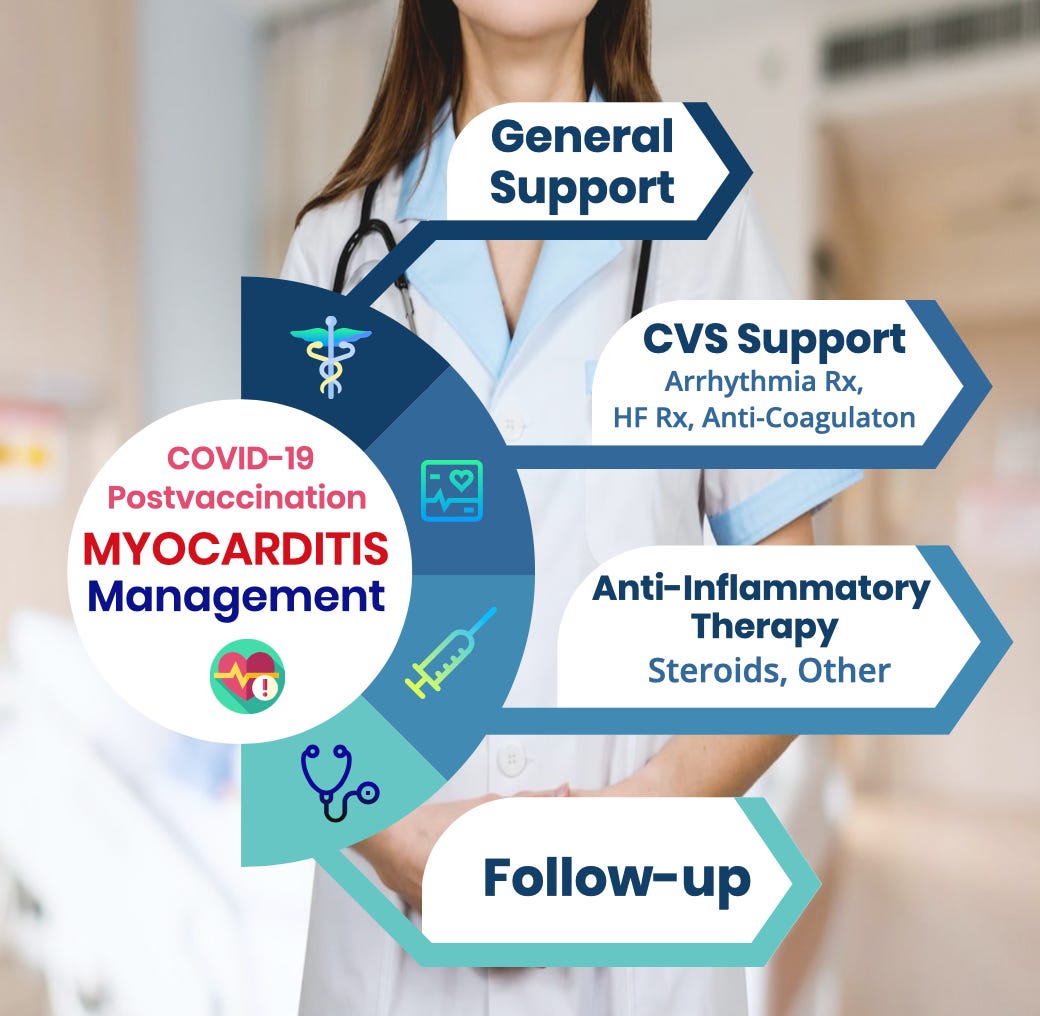
Disclaimer: The information posted here is not to be considered medical advice, and is not intended to replace formal medical consultation. It is not meant to be comprehensive and should be used as a tool to help the user understand and/or assess potential diagnostic and treatment options. It does NOT include all information about conditions, treatments, medications, side effects, or risks that may apply to a specific patient. Patients must speak with a health care provider for complete information about their health, medical questions, and treatment options, including any risks or benefits regarding use of medications. This information does not endorse any treatments or medications as safe, effective, or approved for treating a specific patient. Information Heals and its affiliates disclaim any warranty or liability relating to this information or the use thereof.
To read the Full Disclaimer: Click here | La…
Keep reading with a 7-day free trial
Subscribe to Information Heals to keep reading this post and get 7 days of free access to the full post archives.